Describe Atomic Structure Using the Terms Proton Neutron Electron
In an atom the proton and neutron resides together inside the nucleus. - analyze the structure of the.
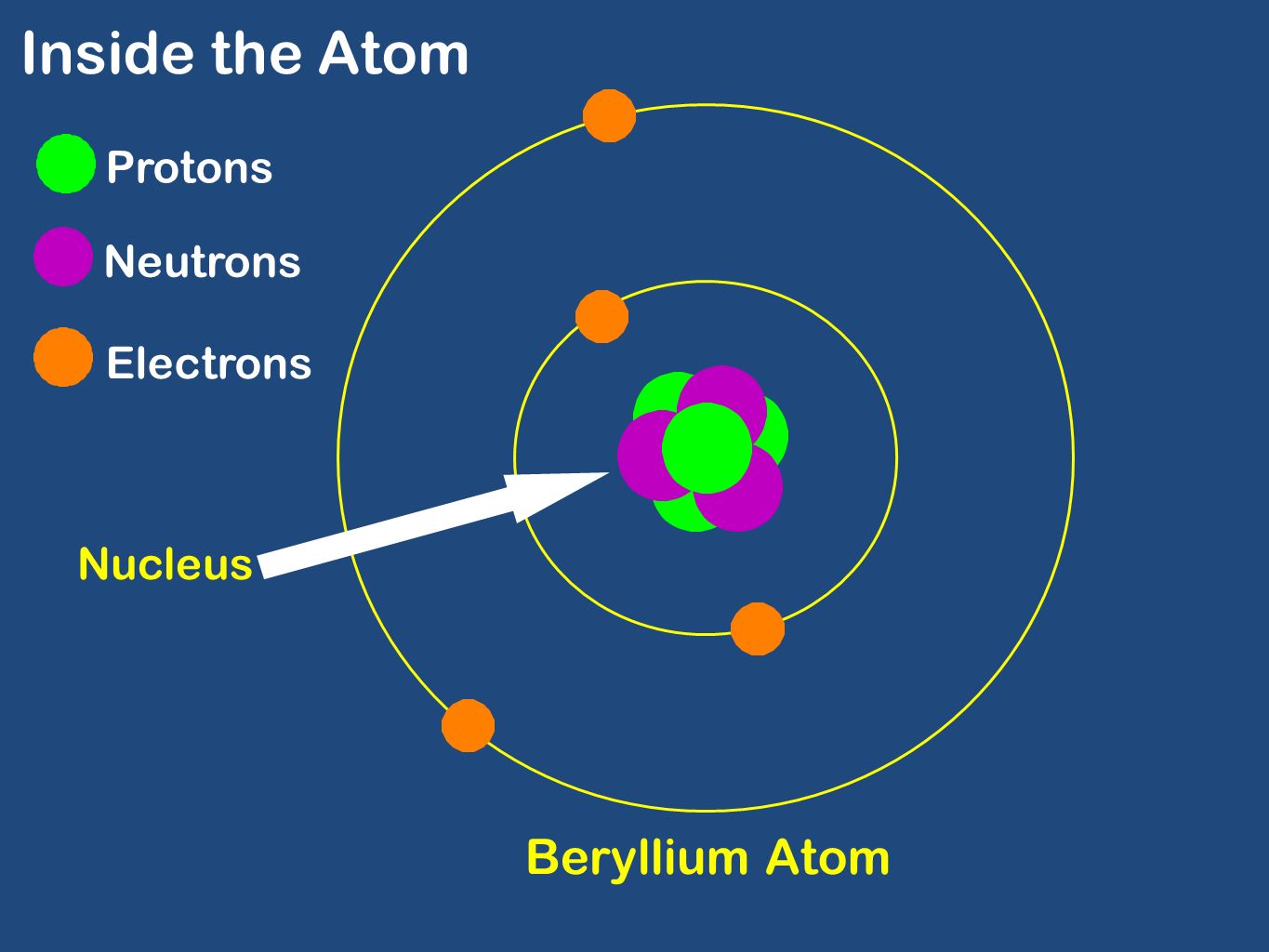
The Atom Structure Inside The Atom Electrons Protons Neutrons Nucleus Beryllium Atom Ppt Download
They are to be found in the space inside the atom but outside the nucleus which is almost the whole of the atom.

. Describe how protons neutrons and electrons relate to atomic structure. Protons and neutrons-make up nucleus each 1 amu Electrons-make. Protons and neutrons form the atomic nucleus.
Research the structure of an atom and draw in your books. Each element has a unique number of protons called the atomic number and when added to the number of neutrons forms a mass number. 2 Nucleon number is the total number of protons and neutrons in an atom.
There are some differences in detail but not in concept. They are also known as Nucleons. The first shell is nearest to the nucleus and can only contain a maximum of two electrons.
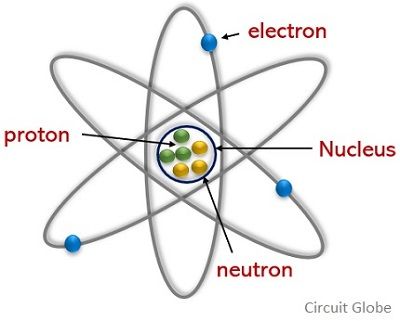
4 In a neutral atom the total number of protons equalsto the total number of electrons. An atom has two major parts in its structure. Neutron is also considered the fundamental particle of an atom.
Things to remember So it is clear that the atom is not the smallest particle. - use atomic mass atomic number and charge to identify neutral atoms ions and isotopes. Protons neutrons and electrons are called subatomic particles because they make up any atom.
Atomic structure The atom consists of three component parts. Neutron An uncharged atomic particle of mass 167493x10-27. 3 Proton number is also known as atomic number while nucleon number is also known as mass number.
Protons and neutrons are arranged into nuclear shells similar to electron shells. Model of the Atom An atom is a building block of matter that cannot be broken apart using any chemical means. Protons are positively charged but they are bonded.
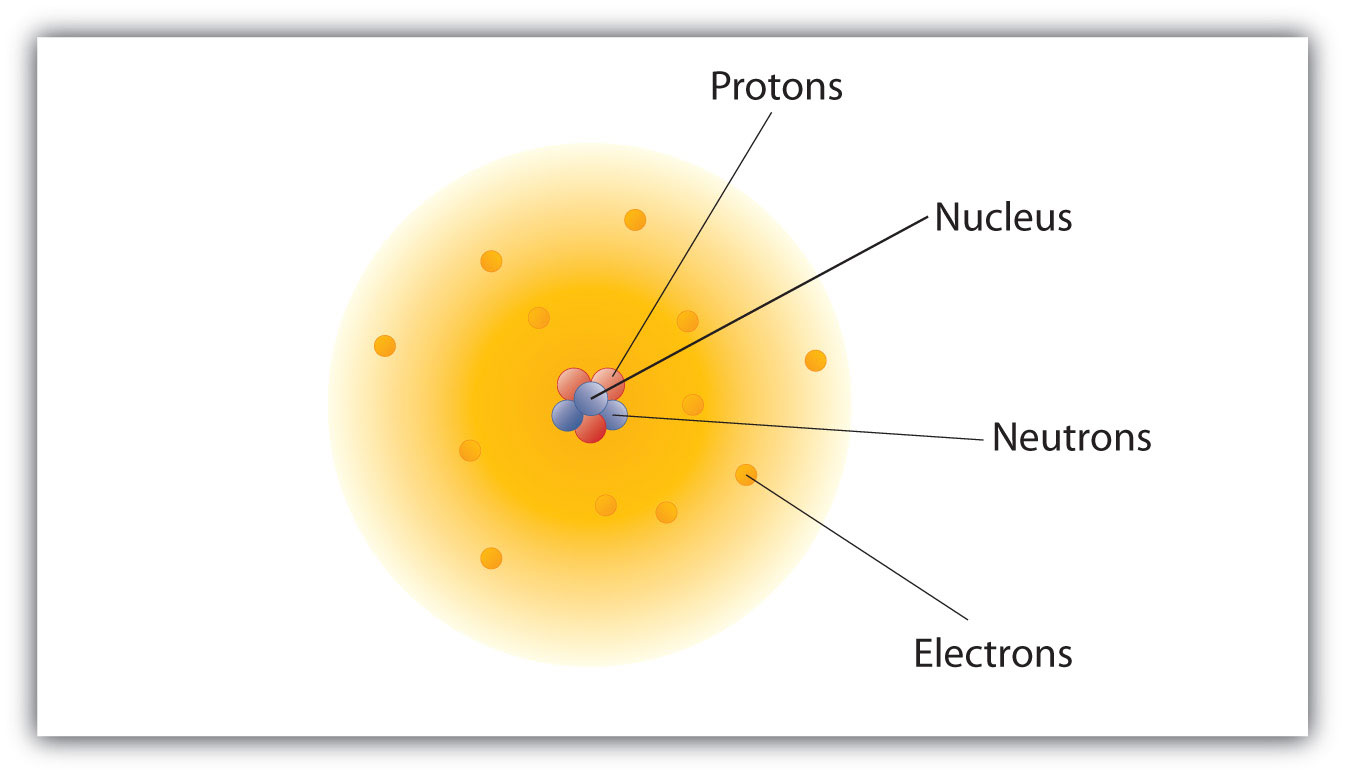
The number of electrons or protons is called the atomic number whereas the sum of protons and neutrons is called atomic mass. Excited state has extra energy and wants to combine isotopseion. The electrons occupy the region around the nucleus.
The energy it takes to remove an electron from an atom. In the example in the diagram for lithium7 73Li is its nuclide atomic notation. Surrounding the nucleus are electrons negative charge in an electron cloud.
When describing an atom you use the atomic number number of protons. The neutron number N mass number protonatomic number In an individual atom the number of protons equals the number of electrons that is the number of positive charges is equal to the number of negative charges to make the atom neutral. If we look again at the neutron structure both the down-quark and the electron occupy the.
- illustrate the structure of the atom by using the Bohr model including the charge relative mass and location of the sub-atomic particles. Thomsons Model of an Atom. The nucleus is composed of neutrons and protons each with a relative mass of 1.
Today they agree that atoms have a positively-charged nucleus made of protons and neutrons and negatively-charged. 1 Proton numberis the total number of protons in an atom. The nucleus is formed by protons and neutrons.
Describe the build-up of electrons in shells and understand the significance of the noble gas electronic structures and of valency electrons. Isotope Atoms with the same number of protons ie. In any atom the number of protons equals the number of.
Same atomic number but a different number of neutrons. Isoelectronic Description for two elemental species with the same electronic configuration. Atomic Structure a state the relative charges and approximate relative masses of a proton a neutron and an electron b describe with the aid of diagrams the structure of an atom as containing protons and neutrons nucleons in the nucleus.
Atomic Structure - describe the characteristics of protons neutrons and electrons in terms of location charge and mass. An atom is composed of protons positive charge and neutrons neutral in an atomic nucleus. The principle quantum number n the orbital angular momentum l and the azimuthal angular momentum or magnetic quantum number m.
Ground is normal and lowest possible state of an. Atomic structure - CCEA Scientists ideas about atoms have changed over time. Its well acknowledged that isolated neutrons will decay into a proton and electron the neutron having a half-life of 103 minutes.
The three dimensional state where an electron can be found is the orbital. The shells are characterized by 3 quantum numbers. Protons Neutrons and Electrons atom Smallest particle of an element mass number The number of protons plus neutrons in the nucleus of an atom atomic number the order of an element in Mendeleevs table of the elements also the number of protons in the elements nucleus.
Okay so a neutron isnt completely stable. It resides in the nucleus. Protons and neutrons are in the nucleus electrons are in the shells Related Posts.
The electrons are present in the three-dimensional space around the nucleus. The particles that contribute to the atoms mass protons and neutrons are contained within a very small central nucleus that has a diameter of about 1 1015m. Structure Bonding Triple quiz Structure Bonding Double quiz.
They are the nucleus and the electron cloud. The atom is made of three fundamental particles. 702 describe the structure of an atom in terms of protons neutrons and electrons and use symbols such as 146C to describe particular nuclei Atoms are made up of protons neutrons and electrons.
Or you can use the mass which is the sum of all neutrons and protons either in atomic mass units or daltons. Social and Applied Aspects.

Atomic Structure Periodic Table Spanish Diagram Quizlet
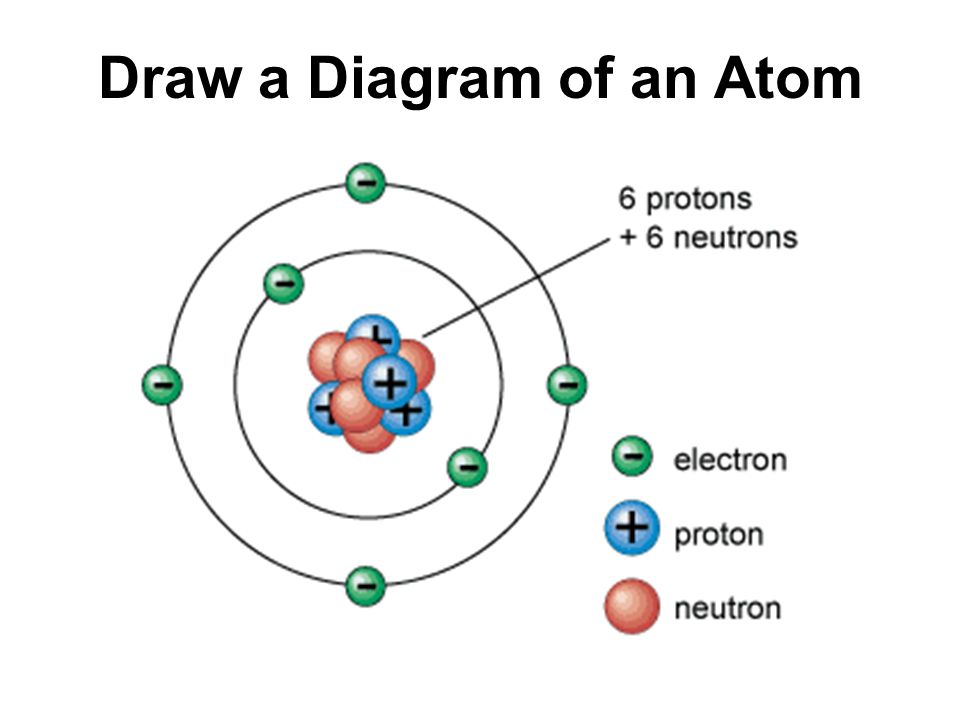
Atomic Structure What Are The 3 Major Parts Of An Atom Proton Neutron Electron Ppt Download

Atomic Structure Nucleus Proton Neutron Electron Mass Charge Isotopes Electron Arrangement Rutherford Bohr Model Of Atom Allotropes History Of Atomic Structure Model Development Ionisation Ions Gcse Chemistry Revision Notes Quizzes Ks4 Science
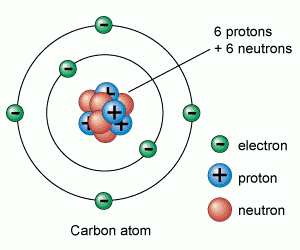
7 02 Describe The Structure Of An Atom In Terms Of Protons Neutrons And Electrons And Use Symbols Such As 146c To Describe Particular Nuclei Tutormyself Chemistry

Introduction To Structure Of Atom Proton Neutron Electron With Examples

Introduction To Structure Of Atom Proton Neutron Electron With Examples

The Structure Of The Atom Boundless Chemistry

Discovery Of Electron Proton And Neutron Who When How
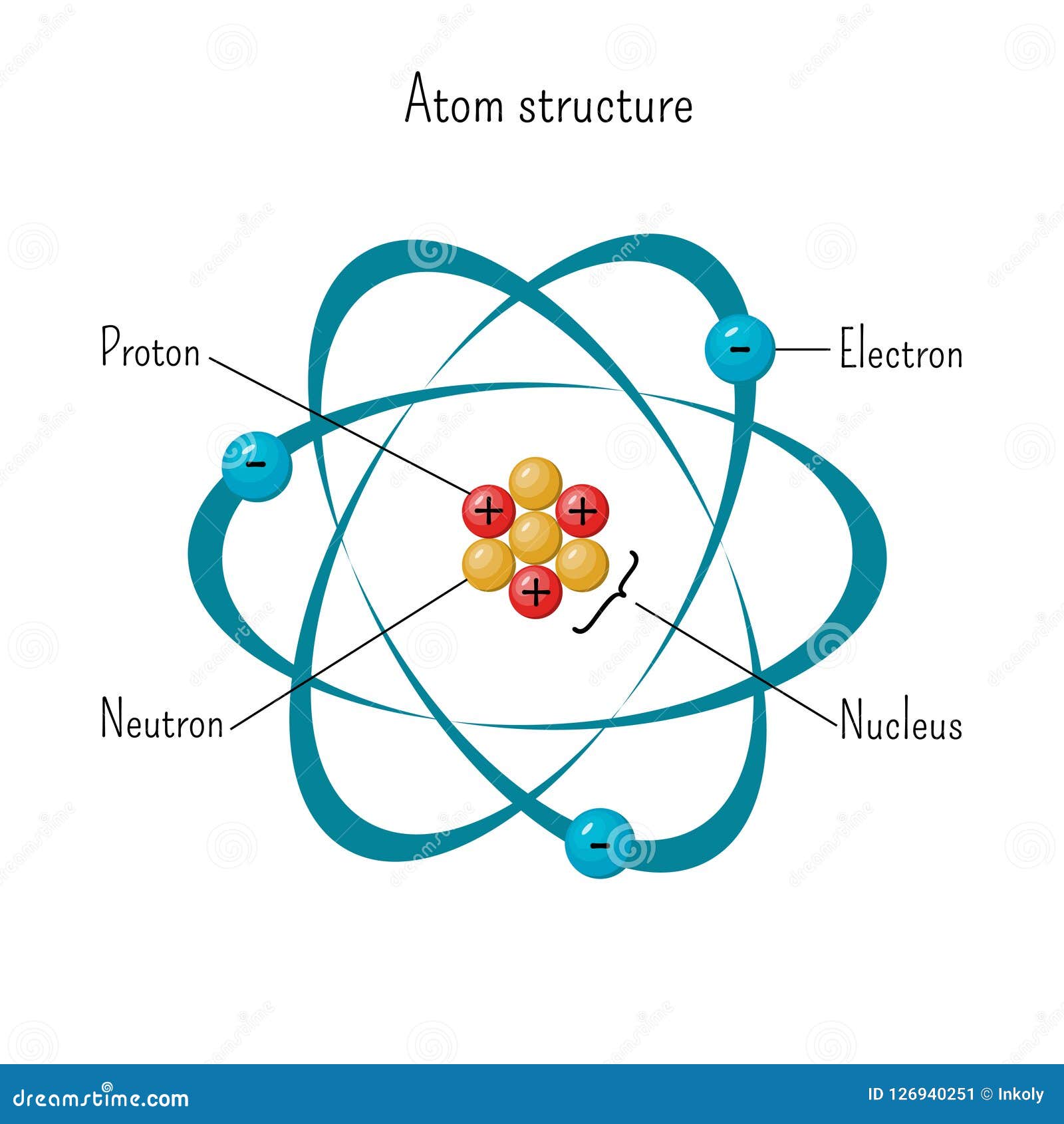
Simple Model Of Atom Structure With Electrons Orbiting Nucleus Of Three Protons And Neutrons Stock Vector Illustration Of Force Diagram 126940251

Ppt 1 1 2 Atomic Structure Describe Protons Neutrons And Electrons Powerpoint Presentation Id 338106
Atomic Structure Wghs Junior Science

Atomic Structure Nucleus Proton Neutron Electron Mass Charge Isotopes Electron Arrangement Rutherford Bohr Model Of Atom Allotropes History Of Atomic Structure Model Development Ionisation Ions Gcse Chemistry Revision Notes Quizzes Ks4 Science

What Is Atomic Structure Definition Meaning And Resources
The Structure Of The Atom Boundless Chemistry

Atomic Structure Electrons Protons Neutrons And Atomic Models
Difference Between Electron And Proton With Comparison Chart Circuit Globe

Comments
Post a Comment